Carbocation

A carbocation is an ion with a positively charged carbon atom. Among the simplest examples are the methenium CH+
3, methanium CH+
5, acylium ions RCO+, and vinyl C
2H+
3 cations.[2]
Until the early 1970s, carbocations were called carbonium ions.[3] In the present-day definition given by the IUPAC, a carbocation is any even-electron cation with significant partial positive charge on a carbon atom. They are further classified in two main categories according to the coordination number of the charged carbon: three in the carbenium ions and five in the carbonium ions. This nomenclature was proposed by G. A. Olah.[4] Carbonium ions, as originally defined by Olah, are characterized by a three-center two-electron delocalized bonding scheme and are essentially synonymous with so-called 'non-classical carbocations', which are carbocations that contain bridging C–C or C–H σ-bonds. However, others have more narrowly defined the term 'carbonium ion' as formally protonated or alkylated alkanes (CR+
5, where R is H or alkyl), to the exclusion of non-classical carbocations like the 2-norbornyl cation.[5]
Definitions
[edit]According to the IUPAC, a carbocation is any cation containing an even number of electrons in which a significant portion of the positive charge resides on a carbon atom.[6] Prior to the observation of five-coordinate carbocations by Olah and coworkers, carbocation and carbonium ion were used interchangeably. Olah proposed a redefinition of carbonium ion as a carbocation featuring any type of three-center two-electron bonding, while a carbenium ion was newly coined to refer to a carbocation containing only two-center two-electron bonds with a three-coordinate positive carbon. Subsequently, others have used the term carbonium ion more narrowly to refer to species that are derived (at least formally) from electrophilic attack of H+ or R+ on an alkane, in analogy to other main group onium species, while a carbocation that contains any type of three-centered bonding is referred to as a non-classical carbocation. In this usage, 2-norbornyl cation is not a carbonium ion, because it is formally derived from protonation of an alkene (norbornene) rather than an alkane, although it is a non-classical carbocation due to its bridged structure. The IUPAC acknowledges the three divergent definitions of carbonium ion and urges care in the usage of this term. For the remainder of this article, the term carbonium ion will be used in this latter restricted sense, while non-classical carbocation will be used to refer to any carbocation with C–C and/or C–H σ-bonds delocalized by bridging.
Since the late 1990s, most textbooks have stopped using the term carbonium ion for the classical three-coordinate carbocation. However, some university-level textbooks continue to use the term carbocation as if it were synonymous with carbenium ion,[7][8] or discuss carbocations with only a fleeting reference to the older terminology of carbonium ions[9] or carbenium and carbonium ions.[10] One textbook retains the older name of carbonium ion for carbenium ion to this day, and uses the phrase hypervalent carbonium ion for CH+
5.[11]
History
[edit]The history of carbocations dates back to 1891 when G. Merling[12] reported that he added bromine to tropylidene (cycloheptatriene) and then heated the product to obtain a crystalline, water-soluble material, C
7H
7Br. He did not suggest a structure for it; however, Doering and Knox[13] convincingly showed that it was tropylium (cycloheptatrienylium) bromide. This ion is predicted to be aromatic by Hückel's rule.
In 1902, Norris and Kehrman independently discovered that colorless triphenylmethanol gives deep-yellow solutions in concentrated sulfuric acid. Triphenylmethyl chloride similarly formed orange complexes with aluminium and tin chlorides. In 1902, Adolf von Baeyer recognized the salt-like character of the compounds formed. The trityl carbocation (shown below) is a stable carbocationic system that has been used as homogeneous organocatalyst in organic synthesis,[14] for example in the form of trityl hexafluorophosphate.[15]
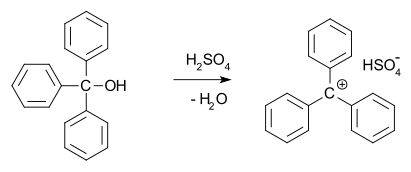
He dubbed the relationship between color and salt formation halochromy, of which malachite green is a prime example.
Carbocations are reactive intermediates in many organic reactions. This idea, first proposed by Julius Stieglitz in 1899,[16] was further developed by Hans Meerwein in his 1922 study[17][18] of the Wagner–Meerwein rearrangement. Carbocations were also found to be involved in the SN1 reaction, the E1 reaction, and in rearrangement reactions such as the Whitmore 1,2 shift. The chemical establishment was reluctant to accept the notion of a carbocation and for a long time the Journal of the American Chemical Society refused articles that mentioned them.
The first NMR spectrum of a carbocation in solution was published by Doering et al.[19] in 1958. It was the heptamethylbenzenium ion, made by treating hexamethylbenzene with methyl chloride and aluminium chloride. The stable 7-norbornadienyl cation was prepared by Story et al. in 1960[20] by reacting norbornadienyl chloride with silver tetrafluoroborate in sulfur dioxide at −80 °C. The NMR spectrum established that it was non-classically bridged (the first stable non-classical ion observed).
In 1962, Olah directly observed the tert-butyl carbocation by nuclear magnetic resonance as a stable species on dissolving tert-butyl fluoride in magic acid. The NMR of the norbornyl cation was first reported by Schleyer et al.[21] and it was shown to undergo proton-scrambling over a barrier by Saunders et al.[22]
Structure and properties
[edit]Carbonium ions
[edit]Carbonium ions can be thought of as protonated alkanes. Although alkanes are usually considered inert, under superacid conditions (e.g., HF·SbF5), the C-H sigma bond can act as a donor to H+. This results in a species that contains a 3c-2e bond between a carbon and two hydrogen atoms, a type of bonding common in boron chemistry, though relatively uncommon for carbon. As an alternative view point, the 3c-2e bond of carbonium ions could be considered as a molecule of H2 coordinated to a carbenium ion (see below). Indeed, carbonium ions frequently decompose by loss of molecular hydrogen to form the corresponding carbenium ion. Structurally, the methanium ion CH+5 is computed to have a minimum energy structure of Cs symmetry. However, the various possible structures of the ion are close in energy and separated by shallow barriers. Hence, the structure of the ion is often described as fluxional. Although there appear to be five bonds to carbon in carbonium ions, they are not hypervalent, as the electron count around the central carbon is only eight, on account of the 3c-2e bond.
Carbenium ions
[edit]At least in a formal sense, carbenium ions are derived from the protonation (addition of H+) or alkylation (addition of R+) of a carbene or alkene. Thus, in at least one of their resonance depictions, they possess a carbon atom bearing a formal positive charge that is surrounded by a sextet of electrons (six valence electrons) instead of the usual octet required to fill the valence shell of carbon (octet rule). Therefore, carbenium ions (and carbocations in general) are often reactive, seeking to fill the octet of valence electrons as well as regain a neutral charge. In accord with VSEPR and Bent's rule, unless geometrically constrained to be pyramidal (e.g., 1-adamantyl cation), 3-coordinate carbon in carbenium ions are usually trigonal planar, with a pure p character empty orbital as its lowest unoccupied molecular orbital (LUMO) and CH/CC bonds formed from C(sp2) orbitals. A prototypical example is the methyl cation, CH+3.
Non-classical ions
[edit]Some carbocations such as the 2-norbornyl cation exhibit more or less symmetrical three-center two-electron bonding. Such structures, referred to as non-classical carbocations, involve the delocalization of the bonds involved in the σ-framework of the molecule, resulting in C–C and C–H bonds of fractional bond order.[23][24] This delocalization results in additional stabilization of the cation. For instance, depicted as a classical carbenium ion, 2-norbornyl cation appears to be a secondary carbocation. However, it is more stable than a typical "secondary" carbocation, being roughly as stable as a tertiary carbocation like t-butyl cation, according to hydride ion affinity.
The existence of non-classical carbocations was once the subject of great controversy. On opposing sides were Herbert C. Brown, who believed that what appeared to be a non-classical carbocation represents the average of two rapidly equilibrating classical species (or possibly two structures exhibiting some degree of bridging or leaning but is nevertheless not symmetric) and that the true non-classical structure is a transition state between the two potential energy minima, and Saul Winstein, who believed that a non-classical structure that possessed a plane of symmetry was the sole potential energy minimum and that the classical structures merely two contributing resonance forms of this non-classical species. George Olah's discovery of superacidic media to allow carbocations to be directly observed, together with a very sensitive NMR technique developed by Martin Saunders to distinguish between the two scenarios, played important roles in resolving this controversy.[25][26] At least for the 2-norbornyl cation itself, the controversy has been settled overwhelmingly in Winstein's favor, with no sign of the putative interconverting classical species, even at temperatures as low as 6 K, and a 2013 crystal structure showing a distinctly non-classical structure.[27][28] A variety of carbocations (e.g., ethyl cation, see above) are now believed to adopt non-classical structures. However, in many cases, the energy difference between the two possible "classical" structures and the "non-classical" one is very small, and it may be difficult to distinguish between the two possibilities experimentally.
See also
[edit]References
[edit]- ^ Scholz, Franziska; Himmel, Daniel; Scherer, Harald; Krossing, Ingo (2013). "Superacidic or Not…︁? Synthesis, Characterisation, and Acidity of the Room-Temperature Ionic Liquid [C(CH3)3]+ [Al2Br7]−". Chemistry – A European Journal. 19 (1): 109–116. doi:10.1002/chem.201203260. PMID 23180742.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 235, ISBN 978-0-471-72091-1
- ^ Robert B. Grossman (2007-07-31). The Art of Writing Reasonable Organic Reaction Mechanisms. Springer Science & Business Media. pp. 105. ISBN 978-0-387-95468-4.
- ^ Olah, George A. (1972). "Stable carbocations. CXVIII. General concept and structure of carbocations based on differentiation of trivalent (classical) carbenium ions from three-center bound penta- of tetracoordinated (nonclassical) carbonium ions. Role of carbocations in electrophilic reactions". Journal of the American Chemical Society. 94 (3): 808–820. doi:10.1021/ja00758a020.
- ^ Sommer, J.; Jost, R. (2000-01-01). "Carbenium and carbonium ions in liquid- and solid-superacid-catalyzed activation of small alkanes". Pure and Applied Chemistry. 72 (12): 2309–2318. doi:10.1351/pac200072122309. ISSN 1365-3075.
- ^ "Carbocation", IUPAC Compendium of Chemical Terminology, International Union of Applied Chemistry, 2009, doi:10.1351/goldbook.C00817, ISBN 978-0967855097, retrieved 2018-11-03
- ^ McMurry, John (August 1999). Organic chemistry (5th ed.). Brooks Cole. ISBN 978-0-534-37617-8.
- ^ Vollhardt, K. Peter C.; Schore, Neil Eric (2018). Organic chemistry: Structure and function (8th ed.). New York. ISBN 9781319079451. OCLC 1007924903.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Yurkanis Bruice, Paula (2004). Organic Chemistry (4th ed.). Pearson/Prentice Hall. ISBN 978-0-13-140748-0.
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. ISBN 978-0-19-850346-0.
- ^ Fox, Marye Anne; Whitesell, James K. (1997). Organic Chemistry. Jones and Bartlett. ISBN 978-0-7637-0413-1.
- ^ Merling, G. (1891). "Ueber Tropin". Berichte der Deutschen Chemischen Gesellschaft. 24 (2): 3108–3126. doi:10.1002/cber.189102402151. ISSN 0365-9496.
- ^ Doering, W. von E.; Knox, L. H. (1954). "The Cycloheptatrienylium (Tropylium) Ion". Journal of the American Chemical Society. 76 (12): 3203–3206. doi:10.1021/ja01641a027.
- ^ "Discovery of an in situ carbocationic system using trityl chloride as a homogeneous organocatalyst". Tetrahedron. 69: 212–218. 2013. doi:10.1016/j.tet.2012.10.042.
- ^ Urch, C. (2001). "Triphenylmethyl Hexafluorophosphate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rt363f. ISBN 0471936235.
- ^ "On the Constitution of the Salts of Imido-Ethers and other Carbimide Derivatives". American Chemical Journal. 21: 101. ISSN 0096-4085.
- ^ Meerwein, H.; Emster, K. van (1922). "About the equilibrium isomerism between bornyl chloride isobornyl chloride and camphene chlorohydrate". Berichte. 55: 2500.
- ^ Rzepa, H. S.; Allan, C. S. M. (2010). "Racemization of Isobornyl Chloride via Carbocations: A Nonclassical Look at a Classic Mechanism". Journal of Chemical Education. 87 (2): 221. Bibcode:2010JChEd..87..221R. doi:10.1021/ed800058c.
- ^ Doering, W. von E.; Saunders, M.; Boyton, H. G.; Earhart, H. W.; Wadley, E. F.; Edwards, W. R.; Laber, G. (1958). "The 1,1,2,3,4,5,6-heptamethylbenzenonium ion". Tetrahedron. 4 (1–2): 178–185. doi:10.1016/0040-4020(58)88016-3.
- ^ Story, Paul R.; Saunders, Martin (1960). "The 7-norbornadienyl carbonium ion". Journal of the American Chemical Society. 82 (23): 6199. doi:10.1021/ja01508a058.
- ^ Schleyer, Paul von R.; Watts, William E.; Fort, Raymond C.; Comisarow, Melvin B.; Olah, George A. (1964). "Stable Carbonium Ions. X.1 Direct Nuclear Magnetic Resonance Observation of the 2-Norbornyl Cation". Journal of the American Chemical Society. 86 (24): 5679–5680. doi:10.1021/ja01078a056.
- ^ Saunders, Martin; Schleyer, Paul von R.; Olah, George A. (1964). "Stable Carbonium Ions. XI.1 The Rate of Hydride Shifts in the 2-Norbornyl Cation". Journal of the American Chemical Society. 86 (24): 5680–5681. doi:10.1021/ja01078a057.
- ^ Strictly speaking, the hyperconjugative stabilization of alkyl-substituted carbocations is a type of three-center bonding. Geometrically, the C–H bonds involved in hyperconjugation are observed (or computed) to "lean" slightly toward the carbocationic center as a result (that is, the +C–C–H bond angle decreases somewhat). Nevertheless, the hydrogen atom is still primarily bonded to the carbon α to the cationic carbon. To qualify as a non-classical carbocation, the two-electron three-center bond needs to feature a group equally (or nearly equally) bonded to two electron-deficient centers. In practice, there is a continuum of possible bonding schemes, ranging from slight involvement of a neighboring group (weak hyperconjugation) to equal sharing of a group between adjacent centers (fully non-classical bonding).
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 447-450. ISBN 9780387448978. OCLC 154040953.
- ^ Olah, George A.; Prakash, G. K. Surya; Saunders, Martin (May 2002). "Conclusion of the classical-nonclassical ion controversy based on the structural study of the 2-norbornyl cation". Accounts of Chemical Research. 16 (12): 440–448. doi:10.1021/ar00096a003.
- ^ George A. Olah - Nobel Lecture
- ^ Yannoni, C. S.; Myhre, P. C.; Webb, Gretchen G. (November 1990). "Magic angle spinning nuclear magnetic resonance near liquid-helium temperatures. Variable-temperature CPMAS spectra of the 2-norbornyl cation to 6 K". Journal of the American Chemical Society. 112 (24): 8991–8992. doi:10.1021/ja00180a060. ISSN 0002-7863.
- ^ Scholz, F.; Himmel, D.; Heinemann, F. W.; Schleyer, P. v R.; Meyer, K.; Krossing, I. (2013-07-05). "Crystal Structure Determination of the Nonclassical 2-Norbornyl Cation". Science. 341 (6141): 62–64. Bibcode:2013Sci...341...62S. doi:10.1126/science.1238849. ISSN 0036-8075. PMID 23828938. S2CID 206549219.
External links
[edit] Media related to Carbocations at Wikimedia Commons
Media related to Carbocations at Wikimedia Commons- Press Release The 1994 Nobel Prize in Chemistry". Nobelprize.org. 9 Jun 2010
